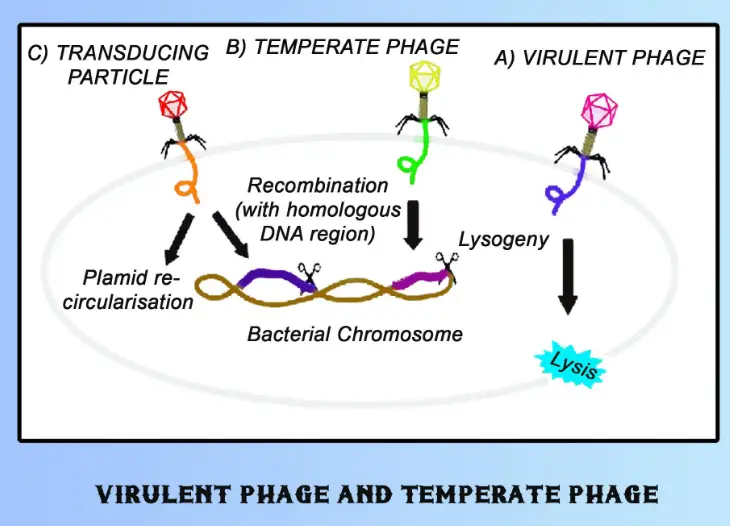
Bacteriophages, commonly referred to as phages, are viruses that infect bacteria. These microscopic entities play a pivotal role in regulating bacterial populations in various environments. Their interactions with bacteria are not only fundamental to microbial ecology but also crucial in biotechnological and medical applications. Phages are broadly categorized into two main types: virulent and temperate, each exhibiting distinct behaviors and life cycles.
Virulent phages are aggressive; they invade bacterial cells, replicate within them, and ultimately cause the host cells to burst, releasing new phage particles. In contrast, temperate phages can adopt a more dormant strategy. They integrate their genetic material into the host’s genome and replicate alongside the host without causing immediate harm. This relationship can switch to a lytic cycle, similar to virulent phages, under specific conditions.
The differences between virulent and temperate phages highlight significant biological mechanisms. These include their infection processes, interactions with host bacteria, and implications for the environment and human health. Understanding these differences is essential for harnessing phages in applications like phage therapy, which could be a vital tool in combating antibiotic-resistant bacterial infections.
Phage Basics
What is a Phage?
Bacteriophages, phages for short, are viruses exclusively infecting bacteria. They are the most abundant biological entities on our planet. Phages play a crucial role in controlling bacterial populations in natural and artificial environments, impacting everything from water ecosystems to human gut health.
Definition and Role
A phage’s primary role is to propagate its genetic material by infecting a bacterium. Through their life cycles, phages influence genetic diversity among bacteria. They contribute to the regulation of bacterial numbers and diversity, which can affect broader ecological dynamics and human health.
Types of Phages
Phages are classified based on their morphology and genetic material. There are two main types based on their lifecycle: virulent and temperate phages. This classification underpins their strategies for reproducing and interacting with host bacteria.
Classification Overview
- Virulent Phages: Follow a lytic cycle, resulting in the destruction of the host cell.
- Temperate Phages: Can undergo a lysogenic cycle, integrating their DNA into the host’s genome, coexisting without causing immediate harm.
Virulent Phages
Characteristics
Virulent phages are characterized by their aggressive approach to replication. They take over the host cell’s machinery quickly and use it to produce new phages.
Lifespan and Behavior
These phages have a short lifespan within a host before they destroy it to release new phage particles. Their behavior is solely focused on replication and the destruction of the host cell.
Mechanism of Action
Infection Process
- Attachment: The phage attaches to the surface of a bacterium.
- Injection: It injects its genetic material into the host.
- Replication: Phage DNA replicates, using the bacterium’s resources.
- Assembly: New phage particles are assembled.
- Lysis: The host cell bursts, releasing new phages.
Impact on Host
Immediate Effects
The immediate effect of a virulent phage infection is the lysis (destruction) of the bacterial cell, which helps control bacterial populations but can also release toxins as bacterial cells break down.
Temperate Phages
Core Traits
Temperate phages can choose between a lytic cycle and a more benign lysogenic cycle, allowing them to either coexist with the host or destroy it.
Lifespan and Dormancy
In the lysogenic cycle, the phage genome integrates into the host cell’s DNA and can remain dormant for extended periods, replicating along with the host’s DNA.
Infection Mechanism
Lysogeny Explained
- Integration: Phage DNA integrates into the host genome.
- Replication: The integrated phage DNA replicates with the host DNA during cell division.
- Induction: Under stress, the phage can initiate a lytic cycle.
Host Interaction
Long-term Effects
The integrated phage DNA can potentially alter the host’s characteristics, including providing resistance to other phages or influencing bacterial virulence.
Comparative Analysis
Lifecycle Differences
Virulent phages strictly follow the lytic cycle, while temperate phages can alternate between lytic and lysogenic cycles based on environmental cues.
Contrasts in Lifecycle Stages
The key difference lies in the presence of a decision phase in temperate phages, absent in virulent phages, dictating whether to kill the host or integrate into its genome.
Genetic Implications
DNA Integration and Consequences
The integration of phage DNA can confer new properties to the host bacteria, such as toxin production or resistance to environmental stresses, impacting bacterial evolution.
Ecological Impact
Effects on Bacterial Populations
Phages influence the composition and behavior of bacterial communities. They can control bacterial overgrowth in environments like the human gut, water bodies, and soil, contributing to ecological balance.
Practical Applications
Medical Use
Phage Therapy Prospects
The resurgence of interest in phage therapy is driven by the escalating crisis of antibiotic resistance. Phage therapy offers a viable alternative, leveraging the natural ability of phages to target and destroy bacteria. This treatment is particularly promising for tackling infections that no longer respond to conventional antibiotics.
Benefits of Phage Therapy:
- Specificity: Phages target specific bacteria, reducing the risk of harming beneficial microflora.
- Adaptability: Phages can evolve with their bacterial targets, potentially overcoming bacterial resistance mechanisms.
Biotechnological Tools
Genetic Engineering and Beyond
Phages carry a toolkit for biotechnological innovation, especially in genetic engineering. Scientists use phages to deliver genetic material into bacteria, an essential technique for genetic manipulation. This capability is pivotal in developing new genetic technologies, from synthetic biology to the creation of genetically modified organisms.
Challenges and Limitations
Phage Resistance
Despite their potential, the use of phages is not without challenges. Bacteria can develop resistance to phages, similar to how they become resistant to antibiotics. Understanding and managing phage resistance is crucial for the sustainable use of phage therapy.
Risks and Management:
- Monitoring: Continuous monitoring of phage effectiveness in clinical settings is essential.
- Phage Cocktails: Using mixtures of phages may help prevent the development of resistance.
Ethical Considerations
The use of phages in healthcare raises ethical questions, particularly regarding safety, regulation, and access.
Usage in Healthcare:
- Regulatory Approval: Navigating the complex regulatory environment is crucial for integrating phage therapy into mainstream medicine.
- Access: Ensuring equitable access to phage-based treatments is a significant ethical consideration.
Future Directions
Research Trends
Ongoing research into phages is uncovering more about their biology, diversity, and application potential. The focus is increasingly on how phages can be integrated into existing and new therapeutic frameworks.
Emerging Studies
Recent studies explore the use of phages in biocontrol—using phages to control harmful bacterial populations in agriculture, food safety, and even waste management. These studies extend the potential of phages beyond human health.
Technological Advancements
Innovations in Phage Application
Technological advancements are propelling the practical application of phages. Innovations in genomic editing, delivery mechanisms, and phage engineering are enhancing the specificity and effectiveness of phages.
Areas of Innovation Include:
- Phage Encapsulation: Improving the delivery and stability of phages in the human body.
- Custom Phage Synthesis: Designing phages specifically tailored to target problematic bacteria.
Frequently Asked Questions
What are bacteriophages?
Bacteriophages are viruses that specifically infect and replicate within bacteria. They are among the most numerous organisms on Earth and play a crucial role in microbial regulation and ecology.
How do virulent phages operate?
Virulent phages follow a lytic cycle where they infect the bacterium, take over its machinery to reproduce, and finally cause the bacterium to lyse (burst), which releases new phages to infect other bacteria.
What defines temperate phages?
Temperate phages can undergo a lysogenic cycle where the phage integrates its DNA into the host’s genome and replicates along with the host cell without causing immediate destruction. This integration can switch to a lytic cycle under certain triggers.
Why are phages important in medicine?
Phages are considered potential tools for phage therapy, an alternative to traditional antibiotics, particularly in treating infections that resist other treatments. They offer a specific, potent way to combat bacterial pathogens.
Can phages contribute to antibiotic resistance?
While phages are a tool against bacterial infections, their interactions with bacteria can sometimes lead to horizontal gene transfer, potentially spreading antibiotic resistance genes among bacterial populations.
Conclusion
The exploration of virulent and temperate phages illuminates a fascinating area of biology that has significant implications for both ecological balance and human health. Their unique mechanisms and behaviors underpin their potential in medical and biotechnological applications, offering hope against the growing threat of antibiotic-resistant bacteria. As research progresses, the nuanced understanding of these phages will likely unlock new pathways for treating diseases and managing microbial populations effectively.