Benzoic acid and sodium benzoate are two compounds frequently encountered in various industrial, pharmaceutical, and food preservation contexts. Their widespread use stems from their effective preservative qualities, safeguarding products from spoilage and extending shelf lives. Though they share a chemical lineage, their properties and implications for use differ significantly, impacting everything from product formulation to regulatory compliance.
Benzoic acid is a naturally occurring compound found in many plants and serves as an important precursor for the synthesis of numerous other chemicals, including sodium benzoate. Sodium benzoate, on the other hand, is the sodium salt of benzoic acid, soluble in water, and used extensively as a food preservative. The key difference lies in their solubility and the environments in which they are most effective; benzoic acid is preferred in more acidic conditions, while sodium benzoate is suitable for a broader range of pH levels.
Both compounds are integral to the food, cosmetic, and pharmaceutical industries, offering unique benefits and challenges. Their roles extend beyond simple preservation, involving nuanced considerations of safety, regulatory adherence, and environmental impact. As the demand for safe and long-lasting products grows, understanding the distinctions between benzoic acid and sodium benzoate becomes crucial for manufacturers and consumers alike.
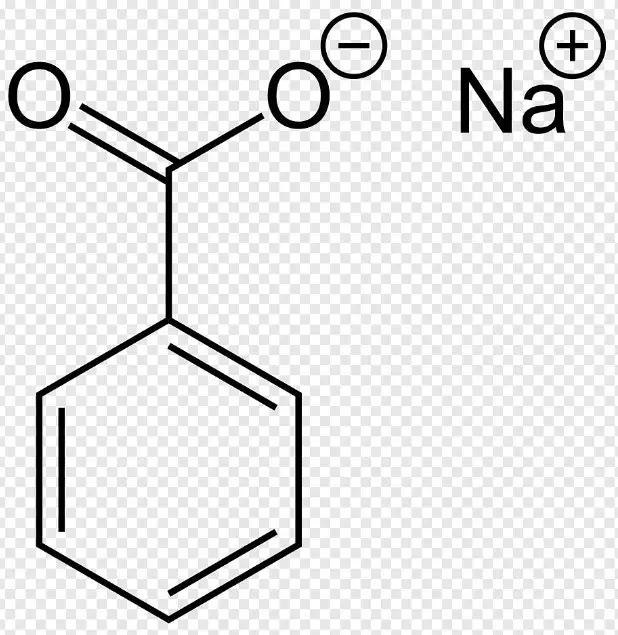
Basics of Benzoic Acid
Definition
Benzoic acid, a simple aromatic carboxylic acid, occurs naturally in many plants and serves as an important precursor for the synthesis of many other organic substances. Its chemical formula is C6H5COOH, indicating a benzene ring attached to a carboxyl group, making it an integral component in the production of various chemical compounds.
Chemical Properties
Benzoic acid is characterized by its mildly acidic nature, making it effective as a preservative. It is less soluble in water but dissolves readily in solvents like ethanol, benzene, and methanol. Upon heating, it sublimates, and its sodium salt, sodium benzoate, is much more soluble in water due to the ionic nature of the salt.
Uses and Applications
This compound finds extensive use across various industries:
- Food industry as a preservative, labeled as E210.
- Pharmaceuticals for its antifungal properties.
- In the manufacture of plastics, as a plasticizer.
- As a part of perfumes and cosmetics for its aromatic properties.
Fundamentals of Sodium Benzoate
Definition
Sodium benzoate, the sodium salt of benzoic acid, has the formula NaC7H5O2. It forms when benzoic acid is dissolved in sodium hydroxide solution, resulting in a substance that is more soluble in water compared to its acid counterpart. This solubility feature significantly broadens its range of applications.
Chemical Properties
Sodium benzoate is known for its high solubility in water and ability to form a clear, colorless solution. It acts as a bacteriostatic and fungistatic agent under acidic conditions by interfering with the microbial energy production processes.
Uses and Applications
Sodium benzoate’s primary uses include:
- A preservative in foods and beverages, especially acidic ones like soft drinks.
- In cosmetics and personal care items to extend shelf life.
- As a corrosion inhibitor in coolants and automotive antifreeze formulations.
Comparative Analysis
Chemical Structure
Benzoic Acid Structure
Benzoic acid features a benzene ring (C6H5) directly linked to a carboxyl group (COOH), a classic example of aromatic carboxylic acid. Its structure is responsible for its aromatic nature and acidic properties.
Sodium Benzoate Structure
Sodium benzoate consists of a benzoate ion (the conjugate base of benzoic acid) and a sodium ion. The presence of the sodium ion increases the compound’s solubility in water significantly.
Key Differences
The key difference lies in the presence of the sodium ion in sodium benzoate, which alters its solubility and reactivity compared to benzoic acid. These structural variations directly impact their applications and effectiveness in various environments.
Solubility
In Water
- Benzoic acid is sparingly soluble in water due to its nonpolar benzene ring.
- Sodium benzoate dissolves well in water, benefiting from the ionic nature of the sodium ion.
In Organic Solvents
Both compounds dissolve in organic solvents, but benzoic acid shows a preference for nonpolar solvents due to its aromatic ring.
Impact of pH
The solubility of benzoic acid in water increases with decreasing pH, as it becomes more protonated. Sodium benzoate’s solubility is less affected by pH changes, remaining consistently high across a broad pH range.
Safety and Regulations
FDA Guidelines
The FDA recognizes both benzoic acid and sodium benzoate as generally recognized as safe (GRAS) when used within prescribed limits in food products.
EU Regulations
In the European Union, both compounds are approved for use as food additives, subject to concentration limits to protect consumer health.
Health Impacts
While generally safe within regulatory limits, excessive intake of these compounds can lead to potential health risks such as metabolic acidosis or allergic reactions in sensitive individuals.
Environmental Impact
Biodegradability
Benzoic acid and sodium benzoate are considered biodegradable under aerobic conditions, breaking down in the environment without accumulating.
Toxicity to Aquatic Life
Both compounds can exhibit toxicity to aquatic organisms at high concentrations, necessitating careful disposal and management practices.
Mitigation Strategies
To reduce environmental impact, industries are encouraged to:
- Minimize release into waterways.
- Treat wastewater effectively.
- Explore greener alternatives with lower ecological footprints.
Applications in Industries
Food Preservation
The role of benzoic acid and sodium benzoate in food preservation cannot be overstated. These compounds extend shelf life by inhibiting the growth of mold, yeast, and bacteria in a wide array of products, including jams, soft drinks, and pickles.
- Role in extending shelf life: These preservatives are added to foods to prevent the growth of harmful microorganisms, thus extending the product’s shelf life and ensuring safety for consumption.
- Common products: You’ll find them in acidic foods and beverages, such as fruit juices, sodas, and condiments, where their efficacy is enhanced by the acidic environment.
- Concentration limits are strictly regulated to ensure safety. Generally, the maximum concentration allowed is 0.1% of the food product’s total weight.
Cosmetics and Personal Care
In the realm of cosmetics and personal care, benzoic acid and sodium benzoate serve as preservatives to prevent the proliferation of microbes, thereby extending product shelf life and maintaining user safety.
- Use in skincare products: These substances are found in products ranging from face creams to shampoos, where they protect against microbial contamination.
- Benefits and concerns: While they effectively preserve products, there is ongoing debate about their sensitivity and potential allergenic effects on some individuals.
- Regulatory compliance is essential. Cosmetic products containing these preservatives must adhere to specific concentration limits and be appropriately labeled to inform consumers.
Pharmaceuticals
In pharmaceuticals, the preservation of drug efficacy and safety is paramount. Benzoic acid and sodium benzoate play crucial roles here as well.
- Benzoic acid in drug formulation: It acts as an excipient, enhancing the overall stability of the drug.
- Sodium benzoate as a preservative: Particularly in liquid medications, it prevents microbial contamination, extending shelf life.
- Clinical implications: The use of these preservatives must balance efficacy with minimizing any potential side effects, especially in medications for sensitive populations.
Consumer Considerations
Label Reading
Empowering consumers through education on label reading is vital. It enables individuals to identify ingredients and understand concentrations, promoting informed choices about the products they purchase.
- Identifying ingredients: Knowing how to spot benzoic acid and sodium benzoate on ingredient lists is crucial for those with sensitivities or concerns.
- Understanding concentrations: Recognizing that these compounds are used within regulated safety limits can alleviate concerns about their presence in products.
- Choosing safer products: For those seeking alternatives, understanding label information helps in selecting products that align with their health and safety preferences.
Alternatives
Exploring alternatives to conventional preservatives can cater to consumer demand for more natural options. Both natural and synthetic preservatives offer comparative effectiveness but have their unique benefits and limitations.
- Natural preservatives: Such as vitamin E (tocopherols) and rosemary extract, offer preservation benefits without synthetic chemicals’ stigma.
- Synthetic options: Include parabens and propylene glycol, which, while effective, face scrutiny over health concerns.
- Comparative effectiveness: While natural preservatives are appealing, they may not always provide the same level of efficacy or shelf life extension as their synthetic counterparts, leading to a careful consideration of the trade-offs involved.
Frequently Asked Questions
What is Benzoic Acid?
Benzoic acid is a colorless crystalline solid with a slightly aromatic odor. It occurs naturally in some fruits and is commonly used in the food industry as a preservative. It works by inhibiting the growth of mold, yeast, and some bacteria, making it an essential component in the shelf-life extension of food products.
How Does Sodium Benzoate Work as a Preservative?
Sodium benzoate is effective as a preservative due to its ability to convert to benzoic acid in acidic conditions. This transformation allows it to inhibit the growth of potentially harmful bacteria, yeast, and molds in food and beverages, ensuring product safety and longevity.
Can Benzoic Acid and Sodium Benzoate Affect Health?
While generally considered safe within regulated concentration limits, excessive intake of benzoic acid and sodium benzoate can lead to adverse health effects. It is crucial to adhere to consumption guidelines established by food safety authorities to minimize any potential risks associated with their use.
Are There Natural Alternatives to These Preservatives?
Yes, there are natural alternatives to benzoic acid and sodium benzoate, including vitamin C (ascorbic acid), rosemary extract, and vinegar. These natural preservatives can offer similar protective benefits in food preservation, though their effectiveness and applicability vary depending on the product.
Conclusion
In the realm of food preservation and safety, the distinction between benzoic acid and sodium benzoate holds significant importance. Both compounds offer vital benefits to the food, pharmaceutical, and cosmetic industries, yet their differences necessitate careful consideration in application. By understanding their unique properties and applications, manufacturers can make informed decisions, ensuring product safety and compliance with regulatory standards.
The choice between benzoic acid and sodium benzoate extends beyond mere technical preference; it encompasses health, environmental, and regulatory dimensions. As consumers become increasingly aware of the ingredients in their products, the demand for transparency and safer alternatives grows. This awareness drives the need for a deeper understanding of preservatives like benzoic acid and sodium benzoate, highlighting the importance of informed choice in the quest for safe and sustainable consumption.

